Learning Objectives
By the end of this section, you will be able to:
- Write and interpret symbols that depict the atomic number, mass number, and charge of an atom or ion
- Define the atomic mass unit and average atomic mass
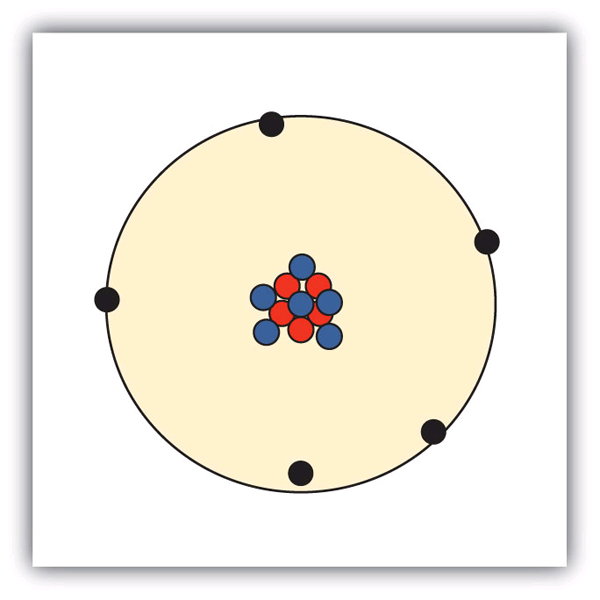
The development of modern atomic theory revealed much about the inner structure of atoms. It was learned that an atom contains a very small nucleus composed of positively charged protons and uncharged neutrons, surrounded by a much larger volume of space containing negatively charged electrons. The nucleus contains the majority of an atom’s mass because protons and neutrons are much heavier than electrons, whereas electrons occupy almost all of an atom’s volume. The diameter of an atom is on the order of 10−10 m, whereas the diameter of the nucleus is roughly 10−15 m—about 100,000 times smaller. For a perspective about their relative sizes, consider this: If the nucleus were the size of a blueberry, the atom would be about the size of a football stadium (Figure 1)!

Atoms—and the protons, neutrons, and electrons that compose them—are extremely small. For example, a carbon atom weighs less than 2 × 10−23 g, and an electron has a charge of less than 2 × 10−19 C (coulomb). When describing the properties of tiny objects such as atoms, we use appropriately small units of measure, such as the atomic mass unit (amu) and the fundamental unit of charge (e). The amu was originally defined based on hydrogen, the lightest element, but now is defined with regard to the most abundant isotope of carbon, atoms of which are assigned masses of exactly 12 amu. The fundamental unit of charge (also called the elementary charge) equals the magnitude of the charge of an electron (e) with e = 1.602 × 10−19 C.
A proton has a mass of 1.0073 amu and a charge of 1+. A neutron is a slightly heavier particle with a mass 1.0087 amu and a charge of zero; as its name suggests, it is neutral. The electron has a charge of 1− and is a much lighter particle with a mass of about 0.00055 amu (it would take about 1800 electrons to equal the mass of one proton.
The number of protons in the nucleus of an atom is its atomic number (Z). This is the defining trait of an element: Its value determines the identity of the atom. A neutral atom must contain the same number of positive and negative charges, so the number of protons equals the number of electrons. Therefore, the atomic number also indicates the number of electrons in an atom. The total number of protons and neutrons in an atom is called its mass number (A). The number of neutrons is therefore the difference between the mass number and the atomic number: A – Z = number of neutrons.
[latex]\begin{array}{r @ {{}={}} l} \text{atomic number (Z)} & \text{number of protons} \\[1em] \text{mass number (A)} & \text{number of protons + number of neutrons} \\[1em] \text{A - Z} & \text{number of neutrons} \end{array}[/latex]
| Name | Location | Charge (C) | Unit Charge | Mass (amu) | Mass (g) |
|---|---|---|---|---|---|
| electron | outside nucleus | −1.602 × 10−19 | 1− | 0.00055 | 0.00091 × 10−24 |
| proton | nucleus | 1.602 × 10−19 | 1+ | 1.00727 | 1.67262 × 10−24 |
| neutron | nucleus | 0 | 0 | 1.00866 | 1.67493 × 10−24 |
| Table 1. Properties of Subatomic Particles | |||||
Atoms are electrically neutral with the same number of positively charged protons and negatively charged electrons. When the numbers of these subatomic particles are not equal, the atom is electrically charged and is called an ion.
Atomic or ionic charge = number of protons − number of electrons
An atom that gains one or more electrons will exhibit a negative charge and is called an anion. Positively charged atoms called cations are formed when an atom loses one or more electrons. For example, a neutral sodium atom (Z = 11) has 11 electrons. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). A neutral oxygen atom (Z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−).
Example 1
Iodine is an essential trace element in our diet; it is needed to produce thyroid hormone. Insufficient iodine in the diet can lead to the development of a goiter, an enlargement of the thyroid gland (Figure 2).
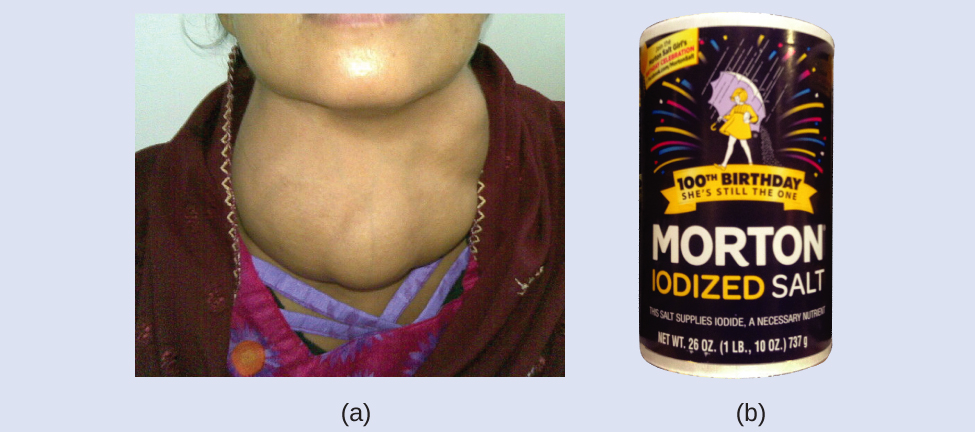
The addition of small amounts of iodine to table salt (iodized salt) has essentially eliminated this health concern in the United States, but as much as 40% of the world’s population is still at risk of iodine deficiency. The iodine atoms are added as anions, and each has a 1− charge and a mass number of 127. Determine the numbers of protons, neutrons, and electrons in one of these iodine anions.
Solution
The atomic number of iodine (53) tells us that a neutral iodine atom contains 53 protons in its nucleus and 53 electrons outside its nucleus. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 − 53 = 74). Since the iodine is added as a 1− anion, the number of electrons is 54 [53 – (1–) = 54].
Test Yourself
An ion of platinum has a mass number of 195 and contains 74 electrons. How many protons and neutrons does it contain, and what is its charge?
Answers
78 protons; 117 neutrons; charge is 4+
Chemical Symbols
A chemical symbol is an abbreviation that we use to indicate an element or an atom of an element. For example, the symbol for mercury is Hg (Figure 3). We use the same symbol to indicate one atom of mercury (microscopic domain) or to label a container of many atoms of the element mercury (macroscopic domain).

The symbols for several common elements and their atoms are listed in Table 2. To avoid confusion with other notations, only the first letter of a symbol is capitalized. All known elements and their symbols are in the periodic table in Figure 2 in Chapter 3.5 The Periodic Table (also found in Appendix A).
| Element | Symbol | Element | Symbol |
|---|---|---|---|
| aluminum | Al | iron | Fe (from ferrum) |
| bromine | Br | lead | Pb (from plumbum) |
| calcium | Ca | magnesium | Mg |
| carbon | C | mercury | Hg (from hydrargyrum) |
| chlorine | Cl | nitrogen | N |
| chromium | Cr | oxygen | O |
| cobalt | Co | potassium | K (from kalium) |
| copper | Cu (from cuprum) | silicon | Si |
| fluorine | F | silver | Ag (from argentum) |
| gold | Au (from aurum) | sodium | Na (from natrium) |
| helium | He | sulfur | S |
| hydrogen | H | tin | Sn (from stannum) |
| iodine | I | zinc | Zn |
| Table 2. Some Common Elements and Their Symbols | |||

Visit this site to learn more about IUPAC, the International Union of Pure and Applied Chemistry, which governs chemical nomenclature (naming). At the IUPAC site you may enjoy exploring their periodic table.
Isotopes
The symbol for a specific isotope of any element is written by placing the mass number as a superscript to the left of the element symbol (Figure 4). The atomic number is sometimes written as a subscript preceding the symbol, but since this number defines the element’s identity, as does its symbol, it is often omitted. For example, magnesium exists as a mixture of three isotopes, each with an atomic number of 12 and with mass numbers of 24, 25, and 26, respectively. These isotopes can be identified as 24Mg, 25Mg, and 26Mg. These isotope symbols are read as “element, mass number” and can be symbolized consistent with this reading. For instance, 24Mg is read as “magnesium 24,” and can be written as “magnesium-24” or “Mg-24.” 25Mg is read as “magnesium 25,” and can be written as “magnesium-25” or “Mg-25.” All magnesium atoms have 12 protons in their nucleus. They differ only because a 24Mg atom has 12 neutrons in its nucleus, a 25Mg atom has 13 neutrons, and a 26Mg has 14 neutrons. Therefore the masses of isotopes of an element, isotopic mass, differ – see Table 3.
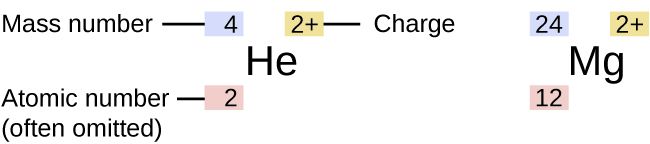
Information about the naturally occurring isotopes of elements with atomic numbers 1 through 10 is given in Table 3. Note that in addition to standard names and symbols, the isotopes of hydrogen are often referred to using common names and accompanying symbols. Hydrogen-2, symbolized 2H, is also called deuterium and sometimes symbolized D. Hydrogen-3, symbolized 3H, is also called tritium and sometimes symbolized T.
| Element | Symbol | Atomic Number | # of Protons | # of Neutrons | Isotopic Mass (amu) | % Natural Abundance |
|---|---|---|---|---|---|---|
| hydrogen | [latex]_1^1\text{H}[/latex]
(protium) |
1 | 1 | 0 | 1.0078 | 99.989 |
| [latex]_1^2\text{H}[/latex]
(deuterium) |
1 | 1 | 1 | 2.0141 | 0.0115 | |
| [latex]_1^3\text{H}[/latex]
(tritium) |
1 | 1 | 2 | 3.01605 | — (trace) | |
| helium | [latex]_2^3\text{He}[/latex] | 2 | 2 | 1 | 3.01603 | 0.00013 |
| [latex]_2^4\text{He}[/latex] | 2 | 2 | 2 | 4.0026 | 100 | |
| lithium | [latex]_3^6\text{Li}[/latex] | 3 | 3 | 3 | 6.0151 | 7.59 |
| [latex]_3^7\text{Li}[/latex] | 3 | 3 | 4 | 7.0160 | 92.41 | |
| beryllium | [latex]_4^9\text{Be}[/latex] | 4 | 4 | 5 | 9.0122 | 100 |
| boron | [latex]_5^{10}\text{B}[/latex] | 5 | 5 | 5 | 10.0129 | 19.9 |
| [latex]_5^{11}\text{B}[/latex] | 5 | 5 | 6 | 11.0093 | 80.1 | |
| carbon | [latex]_6^{12}\text{C}[/latex] | 6 | 6 | 6 | 12.0000 | 98.89 |
| [latex]_6^{13}\text{C}[/latex] | 6 | 6 | 7 | 13.0034 | 1.11 | |
| [latex]_6^{14}\text{C}[/latex] | 6 | 6 | 8 | 14.0032 | — (trace) | |
| nitrogen | [latex]_7^{14}\text{N}[/latex] | 7 | 7 | 7 | 14.0031 | 99.63 |
| [latex]_7^{15}\text{N}[/latex] | 7 | 7 | 8 | 15.0001 | 0.37 | |
| oxygen | [latex]_8^{16}\text{O}[/latex] | 8 | 8 | 8 | 15.9949 | 99.757 |
| [latex]_8^{17}\text{O}[/latex] | 8 | 8 | 9 | 16.9991 | 0.038 | |
| [latex]_8^{18}\text{O}[/latex] | 8 | 8 | 10 | 17.9992 | 0.205 | |
| fluorine | [latex]_9^{19}\text{F}[/latex] | 9 | 9 | 10 | 18.9984 | 100 |
| neon | [latex]_{10}^{20}\text{Ne}[/latex] | 10 | 10 | 10 | 19.9924 | 90.48 |
| [latex]_{10}^{21}\text{Ne}[/latex] | 10 | 10 | 11 | 20.9938 | 0.27 | |
| [latex]_{10}^{22}\text{Ne}[/latex] | 10 | 10 | 12 | 21.9914 | 9.25 | |
| Table 3. Nuclear Compositions of Atoms of the Very Light Elements | ||||||
Example 3
- What is the symbol for an isotope of uranium that has an atomic number of 92 and a mass number of 235?
- How many protons and neutrons are in [latex]_{26}^{56}\text{Fe}[/latex]?
Solution
- The symbol for this isotope is [latex]_{92}^{235}\text{U}[/latex].
- This iron atom has 26 protons and 56 − 26 = 30 neutrons.
Test Yourself
How many protons are in [latex]_{23}^{11}\text{Na}[/latex]?
Answer
11 protons
Example 4
Determine the number of protons, neutrons and electrons for the ion:
![]()
Solution
The atomic number is 17, thus the ion contains 17 protons. The mass number is 35, therefore it contains 35 – 17 = 18 neutrons. Because it is negatively charged (-1), it must have one more electron as compared to protons, thus 17 + 1 = 18 electrons.
Test Yourself
Determine the number of electrons in each of the following ions. Hint: Use the periodic table to first determine the number of protons based on its elemental identity. a) Mg2+ b) Fe3+ c) O2-
Answers
a) 10 b) 23 c) 10

Use this Build an Atom simulator to build atoms of the first 10 elements, see which isotopes exist, check nuclear stability, and gain experience with isotope symbols.
Atomic Mass
Because each proton and each neutron contribute approximately one amu to the mass of an atom, and each electron contributes far less, the atomic mass of a single atom is approximately equal to its mass number (a whole number). However, the average masses of atoms of most elements are not whole numbers because most elements exist naturally as mixtures of two or more isotopes.
The mass of an element shown in a periodic table or listed in a table of atomic masses is a weighted, average mass of all the isotopes present in a naturally occurring sample of that element. This is equal to the sum of each individual isotope’s mass multiplied by its fractional abundance.
Example 6
A meteorite found in central Indiana contains traces of the noble gas neon picked up from the solar wind during the meteorite’s trip through the solar system. Analysis of a sample of the gas showed that it consisted of 91.84% 20Ne (mass 19.9924 amu), 0.47% 21Ne (mass 20.9940 amu), and 7.69% 22Ne (mass 21.9914 amu). What is the average mass of the neon in the solar wind?
Solution
The average mass of a neon atom in the solar wind is 20.15 amu. (The average mass of a terrestrial neon atom is 20.1796 amu. This result demonstrates that we may find slight differences in the natural abundance of isotopes, depending on their origin.)
Test Yourself
A sample of magnesium is found to contain 78.70% of 24Mg atoms (mass 23.98 amu), 10.13% of 25Mg atoms (mass 24.99 amu), and 11.17% of 26Mg atoms (mass 25.98 amu). Calculate the average mass of a Mg atom.
Answer
24.31 amu

Visit this site to make mixtures of the main isotopes of the first 18 elements, gain experience with average atomic mass, and check naturally occurring isotope ratios using the Isotopes and Atomic Mass simulation.
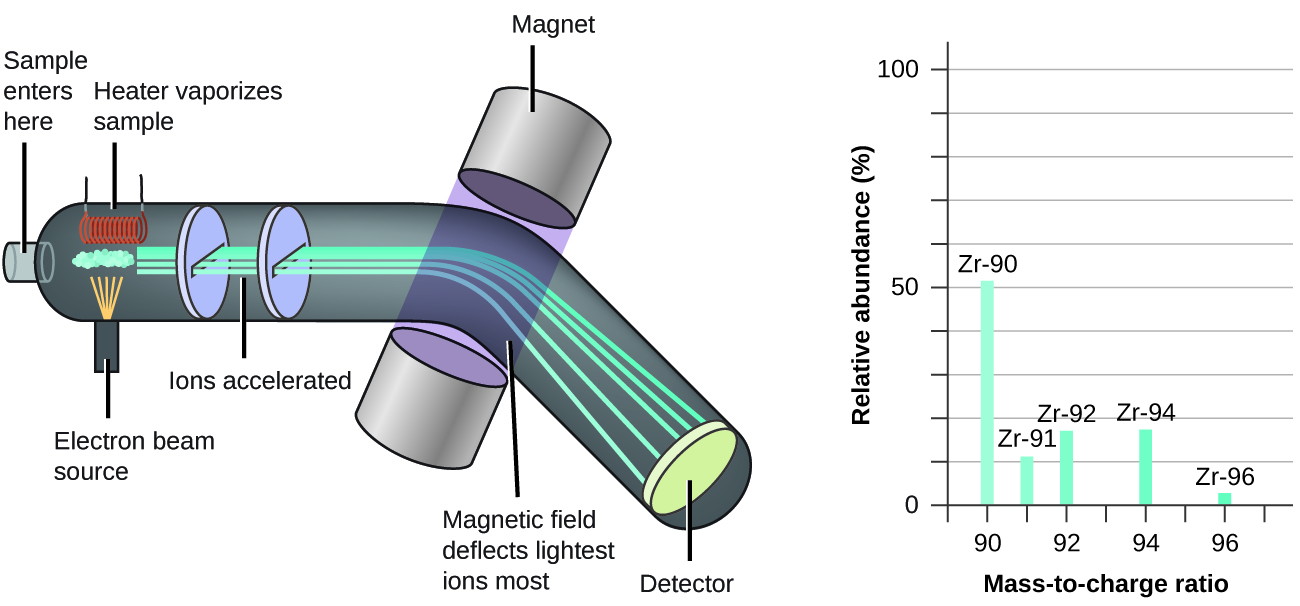
The occurrence and natural abundances of isotopes can be experimentally determined using an instrument called a mass spectrometer. Mass spectrometry (MS) is widely used in chemistry, forensics, medicine, environmental science, and many other fields to analyze and help identify the substances in a sample of material. A mass spectrum is a plot of the relative number of ions generated versus their mass-to-charge ratios, with the height of each vertical feature or peak in a mass spectrum proportional to the fraction of particles with the specified ratio in the sample. Mass spectrometry can thus show the relative abundances of specific isotopes in a sample, directly.
Key Concepts and Summary
An atom consists of a small, positively charged nucleus surrounded by electrons. The nucleus contains protons and neutrons; its diameter is about 100,000 times smaller than that of the atom. The mass of one atom is usually expressed in atomic mass units (amu), which is referred to as the atomic mass.
Protons are particles with a charge of 1+ and a mass of 1.0073 amu. Neutrons are particles with no charge and a mass of 1.0087 amu. Electrons are particles with a charge of 1− and a mass of 0.00055 amu. The number of protons in the nucleus is called the atomic number (Z) and is the property that defines an atom’s elemental identity. The sum of the numbers of protons and neutrons in the nucleus is called the mass number and, expressed in amu, is approximately equal to the mass of the atom. An atom is neutral when it contains equal numbers of electrons and protons.
Isotopes of an element are atoms with the same atomic number but different mass numbers; isotopes of an element, therefore, differ from each other only in the number of neutrons within the nucleus. When a naturally occurring element is composed of several isotopes, the atomic mass of the element represents the average of the masses of the isotopes involved. A chemical symbol identifies the atoms in a substance using symbols, which are one-, two-, or three-letter abbreviations for the atoms.
Key Equations
- [latex]\displaystyle{} \text{average mass} = \sum_{i} (\text{fractional abundance} \times \text{isotopic mass})_i[/latex]
Review-Reflect, Extend
Review-Reflect
1. Write the symbol for each of the following ions:
a) the ion with a 1+ charge, atomic number 55, and mass number 133
b) the ion with 54 electrons, 53 protons, and 74 neutrons
c) the ion with atomic number 15, mass number 31, and a 3− charge
d) the ion with 24 electrons, 30 neutrons, and a 3+ charge
2. Open the Build an Atom simulation and click on the Atom icon.
a) Pick any one of the first 10 elements that you would like to build and state its symbol.
b) Drag protons, neutrons, and electrons onto the atom template to make an atom of your element.
State the numbers of protons, neutrons, and electrons in your atom, as well as the net charge and mass number.
c) Click on “Net Charge” and “Mass Number,” check your answers to (b), and correct, if needed.
3. The following are properties of isotopes of two elements that are essential in our diet. Determine the number of protons, neutrons and electrons in each and name them.
a) atomic number 26, mass number 58, charge of 2+
b) atomic number 53, mass number 127, charge of 1−
4. Sketch a diagram of a boron atom, which has five protons and six neutrons in its nucleus.
5. Define atomic number. What is the atomic number for a boron atom?
6. Which pair represents isotopes?
a) ![]() b) 26F and 25M c) 14S and 15P
b) 26F and 25M c) 14S and 15P
7. Give complete symbols of each atom, including the atomic number and the mass number.
a) an oxygen atom with 8 protons and 8 neutrons
b) a potassium atom with 19 protons and 20 neutrons
c) a lithium atom with 3 protons and 4 neutrons
8. Americium-241 is an isotope used in smoke detectors. What is the complete symbol for this isotope?
9. Give atomic symbols for each element.
a) sodium b) argon c) nitrogen d) radon
10. Give the name of the element.
a) F b) Cl c) Br d) I
Extend
1. Average atomic masses listed by IUPAC are based on a study of experimental results. Bromine has two isotopes 79Br and 81Br, whose masses (78.9183 and 80.9163 amu) and abundances (50.69% and 49.31%) were determined in earlier experiments. Calculate the average atomic mass of bromine based on these experiments.
Answers to Review-Reflect
1. (a) 133Cs+; (b) 127I−; (c) 31P3−; (d) 57Co3+
2. (a) Carbon-12, 12C; (b) This atom contains six protons and six neutrons. There are six electrons in a neutral 12C atom. The net charge of such a neutral atom is zero, and the mass number is 12. (c) The preceding answers are correct. (d) The atom will be stable since C-12 is a stable isotope of carbon. (e) The preceding answer is correct. Other answers for this exercise are possible if a different element of isotope is chosen.
3. (a) Iron, 26 protons, 24 electrons, and 32 neutrons; (b) iodine, 53 protons, 54 electrons, and 74 neutrons
5. The atomic number is the number of protons in a nucleus. Boron has an atomic number of five.
6. (a)
7. a) [latex]_{8}^{16}\text{O}[/latex] b) [latex]_{19}^{39}\text{K}[/latex] c) [latex]_{3}^{7}\text{Li}[/latex]
8. [latex]_{95}^{241}\text{Am}[/latex]
9. a) Na b) Ar c) N d) Rn
10. a) fluorine b) chlorine c) bromine d) iodine
Glossary
electron: negatively charged, subatomic particle of relatively low mass located outside the nucleus
anion: negatively charged atom or molecule (contains more electrons than protons)
atomic mass: average mass of atoms of an element, expressed in amu
atomic mass unit (amu): (also, unified atomic mass unit, u, or Dalton, Da) unit of mass equal to [latex]\frac{1}{12}[/latex] of the mass of a 12C atom
atomic number (Z): number of protons in the nucleus of an atom
cation: positively charged atom or molecule (contains fewer electrons than protons)
chemical symbol: one-, two-, or three-letter abbreviation used to represent an element or its atoms
Dalton (Da): alternative unit equivalent to the atomic mass unit
fundamental unit of charge: (also called the elementary charge) equals the magnitude of the charge of an electron (e) with e = 1.602 × 10−19 C
ion: electrically charged atom or molecule (contains unequal numbers of protons and electrons)
isotopic mass: mass of an isotope of an element, expressed in amu
mass number (A): sum of the numbers of neutrons and protons in the nucleus of an atom
unified atomic mass unit (u): alternative unit equivalent to the atomic mass unit