15
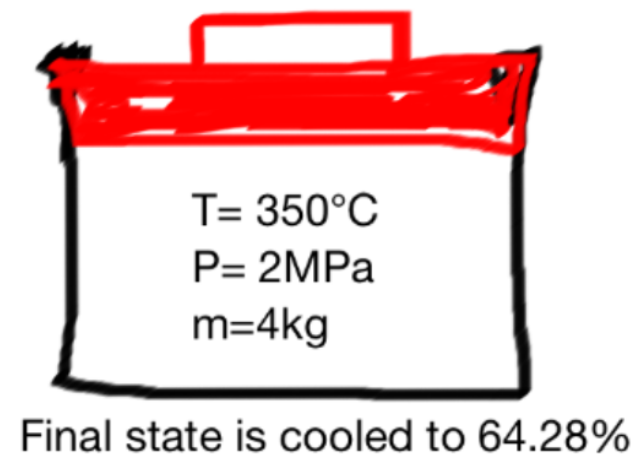
A movable cover is located at the top of an opened metal box. The metal box contains steam at 350![]() C, and pressure of 2 MPa. Currently the steam is about to be cooled. Determine the compression work
C, and pressure of 2 MPa. Currently the steam is about to be cooled. Determine the compression work ![]() for the steam and pressure and its final temperature where the mass of the movable box and metal box is 4kg.
for the steam and pressure and its final temperature where the mass of the movable box and metal box is 4kg.
![]()
![]()
![]()
![]()
Given:
![]()
![]()
![]()
![]()
![]()
Find:
Determine the compression work ![]() for the steam and pressure and its final temperature.
for the steam and pressure and its final temperature.
Solution:
Using table A-6
At First State
![]()
![]()
![]()
At Final State
![]()
![]()
![]()
a) the compression work
![]()
![]()
b) the volume of the metal box at the final state is 64.28% from its initial volume, so the work will be as
![]()
![]()
![]()
![]()
![]()