22
Question 1
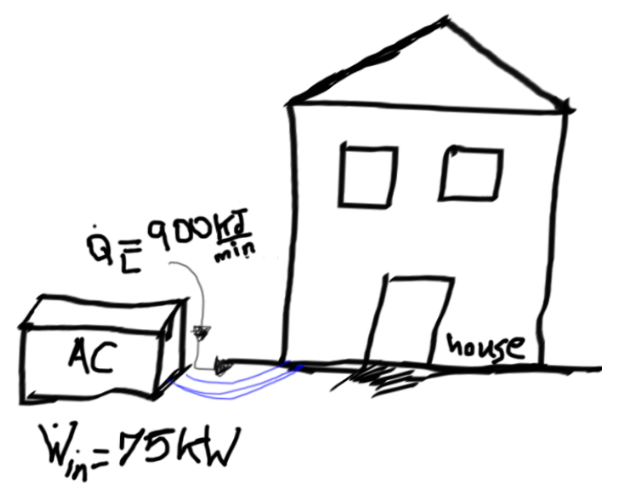
A cooling system is being used in order to decrease heat gradually from a house at a rate of 900 kJ/min while drawing electric power at a rate of 75 kW. Determine the COP of this system and the rate of heat transfer to the outside weather.
Given:
![]()
![]()
Find:
Determine the COP of this system and the rate of heat transfer for the outside weather.
Solution:
The COP of this system
![]()
The rate of heat transfer for the outside weather.
![]()
Question 2
Through the isothermal heat addition process of Carnot cycle, amount of hear, 750 kJ, was added to a fluid from a source at ![]() C. Determine the entropy change of the working fluid, the entropy change of the source, and the total entropy change for the process.
C. Determine the entropy change of the working fluid, the entropy change of the source, and the total entropy change for the process.
Given:
![]()
![]()
Find:
Determine the entropy change of the working fluid, the entropy change of the source, and the total entropy change for the process.
Solution:
![]()
![]()
![]()